
From Biology to Modeling
- Discovery
- Preclinical
- Clinical Ph1
- Clinical Ph2
- Clinical Ph3
- Efficacy
- Balanced
- safety
Episode 3
Donec ornare tortor ultrices mi dictum
From Biology to Modeling
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium.
At a glance: TCEs outside blood, key hurdles: target choice, immune access, and spatial activation
- Reduced need for extensive animal testing
- Earlier identification of potential drug candidates
- More accurate prediction of drug efficacy and safety in humans
- Optimization of dosing regimens
- Better understanding of drug-drug interactions
Why TCEs struggle outside the blood
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium. Lorem ipsum dolor sit amet, consectetur adipiscing elit.Lorem ipsum dolor sit amet, consectetur adipiscing elit. Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusantium doloremque laudantium. Lorem ipsum dolor sit amet, consectetur adipiscing elit.
References
- Peterson, M.C., Riggs, M.M. (2010). Applications of quantitative systems pharmacology in model-informed drug development. Clinical Pharmacology & Therapeutics, 93(5), 426–435.
- Zhang, L., Pfister, M., Meibohm, B. (2021). Concepts and challenges in quantitative pharmacology and model-based drug development. AAPS Journal, 23(2), 77.
- Johnson, T.N., Rostami-Hodjegan, A. (2020). Physiologically based pharmacokinetic modeling in drug development and regulatory science.Clinical Pharmacology & Therapeutics, 107(1), 40–47.
- Liu, A., Bassingthwaighte, J., Sorensen, A. (2020). Integrating artificial intelligence with QSP models: Current status and future directions. CPT: Pharmacometrics & Systems Pharmacology, 10(7), 654–666.
You may be looking for other services

FIH dose for mRNA-based Cancer Vaccine
Can we identify recommended FIH dose and regimen for novel mRNA-based checkpoint vaccine?
Read More →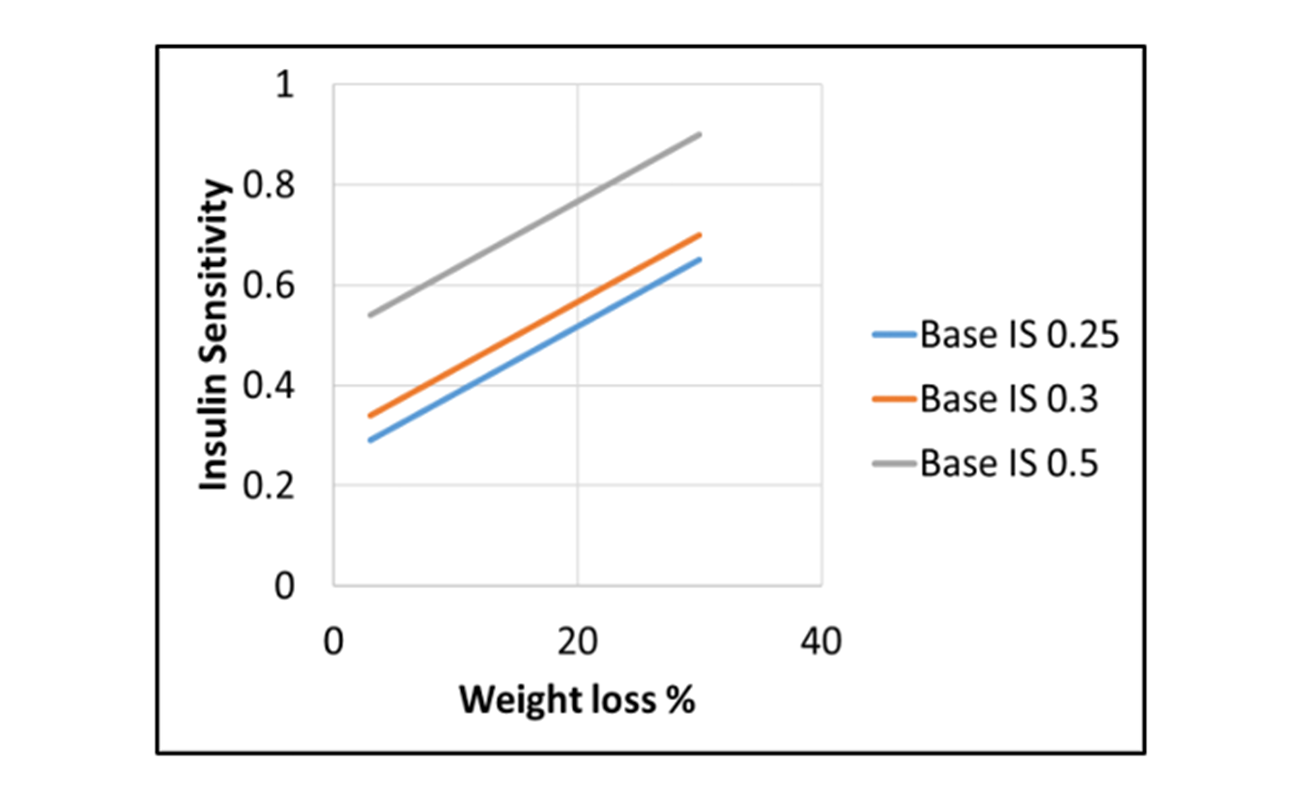
Hypoglycemia exacerbation in T2Ds who exhibit weight loss on insulin Rx
Does hypoglycemia increase when anti-hyperglycemic agents are administered to Type 2 Diabetes patients undergoing weight…
Read More →
QSP in R
Author: Anuraag Saini What do we do? At Vantage Research, we assist decision-making throughout the…
Read More →Meet Dr. Bhairaj Paleja, PhD


